Electrical Engineering ⇒ Topic : Chemical Changes During Discharging
|
|
| Sachin
| |
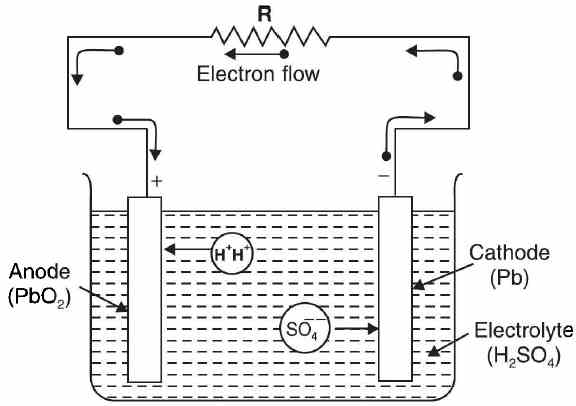
Chemical Changes During Discharging By discharging of a cell, we mean that it is delivering current to the external circuit. Consider a charged lead acid cell with anode of Pb02 and cathode of Pb; the electrolyte being dilute H2SO[See Fig. (a)]. Sulphuric acid splits up into hydrogen ions (H+H+) and sulphate ions (SO4--).The sulphate ions move towards the cathode and hydrogen ions move towards the anode causing the following chemical reactions Figure (a) At cathode. On reaching the cathode, a sulphate ion (SO4- -) gives up its two extra electrons to become sulphate radical. These electrons given up at the cathode move through the external circuit to the anode where they are available to neutralise the positive ions (H+H+) arriving there. Since sulphate radical cannot exist, it enters into chemical reaction with cathode material (Pb) to form lead sulphate (PbS04).
As the cell delivers cun-ent, both the plates start getting converted into lead sulphate (PbS04).The water produced in the chemical reactions above dilutes the electrolyte (H2S04) and lowers its specific gravity. When the specific gravity (which can be measured by a hydrometer) of the electrolyte falls to 1.18, the cell is fully discharged. The chemical changes that take place during discharging of a lead-acid cell can be summed up as under
It is important to note that e.m.f. of the cell provides little indication to the state of discharge of the cell since it remains close to 2 V for 90% of the discharge period. In practice, specific gravity of the electrolyte (H2SO4) is used to know the state of discharge. The cell should be recharged when specific gravity of H2S04 falls to about 1.18. | |
|
| |
!! OOPS Login [Click here] is required for more results / answer