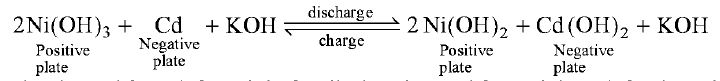Electrical Engineering ⇒ Topic : Nickel-Cadmium Cell
|
|
| Maninder
| |
Nickel-Cadmium Cell Nickel-Cadmium cell was developed by Waldermar Junger, a Swede, in 1899. When the nickelcadmium cell is in the charged condition, the active material on the positive plate is Ni(OH)3 and that on the negative plate is cadmium (Cd). The electrolyte is the same as in the nickel-iron cell i.e. potassium hydroxide (KOH). When the cell is discharged, the positive plate is converted to Ni(OH)2 and the negative plate to Cd (OH)2. When the cell is recharged, the chemical process is reversed i.e. positive plate is converted to Ni(OH)3 and negative plate to Cd. The equation of chemical reaction is
The equation is read from left to right for discharging and from right to left when charging. The following points may be noted in these electro-chemical reactions There is merely transfer of hydroxyl ions (20H -) from one plate to the other. Thus during discharging, hydroxyl ions from the positive plate are transferred to the negative plate to form Cd (OH)2. The reverse happens during charging. The specific gravity of the electrolyte (KOH) remains unchanged during charge or discharge. The e.m.f. of a nickel-cadmium cell is about 1.2 V when charged and falls to 1.1 V when discharged. Since the specific gravity of the electrolyte (KOH) remains unchanged during charge or discharge, it cannot be used as an indication to the state of charge of the cell. Instead, a voltmeter is employed to determine whether the cell is charged upto its rated voltage | |
|
| |
!! OOPS Login [Click here] is required for more results / answer