Electrical Engineering ⇒ Topic : Practical Applications of Electrolysis
|
|
| Sachin
| |
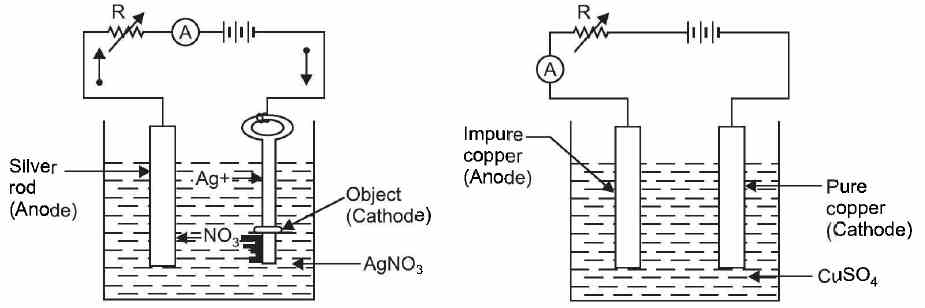
Practical Applications of Electrolysis The phenomenon of electrolysis has many industrial and commercial applications. A few of them are discussed below by way of illustration. (1) Electroplating. The process of depositing a thin layer of superior metal (e.g., gold, silver, nickel, etc.) over an inferior metal (e.g., iron) is known as electroplating. The aim of electroplating is to provide good appearance and protect the object against corrosion. Fig. (a) shows a simple arrangement of silver plating. The object to be plated (e.g. a small iron key) is used as the cathode.The metal to be deposited (silver in this case) is made the anode. The solution of compound of the metal to be deposited in this case AgNO3 is taken as the electrolyte. When d.c. supply is applied to the electrodes, the silver rod (i.e. anode) continuously dissolves into the solution and gets deposited on the cathode (i.e. the object).
(a) (b) figure (2) Refining of metals. Many metals are purified by the process of electrolysis. The impure metal is used as the anode and a small piece of pure metal is used as the cathode. The salt solution of the metal to be purified is used as the electrolyte. Fig.(b) shows a simple arrangement for electrolytic refining of *copper. When d.c. supply is applied to the electrodes, pure copper from anode goes into the solution and gets deposited on the cathode. The impurities sink to the bottom of the container and from an 'anode mud'. Similarly, electrolytic process can be employed for refining other metals such as silver, gold, platinum and nickel. (3) Production of chemicals. The process of electrolysis is being extensively used for the commercial production of chemicals like sodium carbonate, sodium bicarbonate, caustic soda etc. (4) Extraction of metals from ores. Many metals such as Al, Mg, Zn, Cu, etc. are extracted from their ores by electrolysis.Production of oxygen and hydrogen. By the electrolysis of acidic water, oxygen and hydrogen can be produced. (5) Electrolytic capacitor. An electrolytic capacitor consists of two aluminium foils in which a very thin dielectric is sandwitched by the process of electrolysis. Two foils of aluminium (which act as electrodes) are dipped in a solution of boric acid, glycerine and ammonia water. When current is passed between anode and cathode, a very thin film of aluminium oxide (Al203) is formed on the anode surface. This oxide film acts as a dielectric. Since the oxide film is very thin, the electrolytic capacitor has a very large capacitance. (6) Anodising. It is the process of coating aluminium oxide on the aluminium itself by electrolysis to protect it against corrosion. The aluminium article is made the anode and dil. H2SO4 is the electrolyte. (7) Electro-typing. This is the process in which we obtain an impression of a page of the book in **copper. The ordinary type is an-anged to form a page of the book. Then a special kind of wax ispoured over it. When the wax hardens and is peeled off, it bears an impression of that page. The waxmould is uniformly coated with a very thin layer of powdered graphite to make it a conductor. The mould is made the cathode and a pure copper plate as the anode. The two electrodes are immersed in a bath containing copper sulphate solution (CuSO4) as the electrolyte. When d.c. supply is applied to the electrodes, copper from anode goes into the solution and gets deposited over the cathode i. e. wax-mould. When sufficient thick layer (as thick as ordinary visiting card) of copper is deposited on the cathode (i.e. wax-mould), the circuit is disconnected. The wax is removed from the cathode and the copper plate obtained would have an impression of the page in copper. Several hundred thousands impressions of the page can be taken from this plate | |
|
| |
!! OOPS Login [Click here] is required for more results / answer